Mild metabolic acidosis is extremely common for those who follow a traditional western style diet, particularly when ageing and chronic disease states are added to the mix. This has implications for bone health as minerals are pulled from the bone to maintain the blood’s pH strictly between 7.35 and 7.45.1 At the other end of the spectrum, should the blood become too alkaline, the kidneys remove those minerals from the blood and they are excreted, potentially leading to increased renal load.
Is osteoporosis a lack of calcium or over-acidity?
A randomised, double blind, placebo-controlled trial that gave potassium citrate to a group of postmenopausal women for one year aimed to investigate the association between acid load and bone loss. It showed a significant decrease in bone turnover, starting in the first month of treatment.2 This decrease was clearly associated with potassium citrate treatment and is consistent with the hypothesis that metabolic acidosis increases osteoclast activity.2
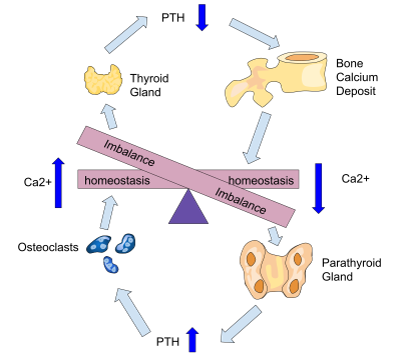
The role of the thyroid glands:
Parathyroid (PTH) hormone regulates calcium homeostasis as part of a tightly controlled system. PTH plays an important role in mobilising calcium from its primary storage in the skeleton and by increasing calcium absorption from the intestine by increasing the kidney’s synthesis of calcitriol (1,25-dihydroxyvitamin D). PTH also increases calcium re-absorption from the kidneys and facilitates the excretion of phosphorus.9
Citrate minerals, including magnesium citrate, are easily and readily metabolised with less dependence on adequate stomach pH than other forms, including magnesium oxide.3 They also have positive effects on bone health via their alkalising properties.
Supplemental carbonate minerals, in contrast, react in the stomach to produce carbon dioxide, which is immediately released, a chloride salt and water. Calcium carbonate, for example, has a very strong gastric pH neutralising effect and can produce acid rebound. In contrast, sodium bicarbonate is a gentle, short-acting antacid with little to no adverse effects.4,5
Very little or no carbonates make it into systemic circulation to act as buffering agents in the blood. Most of the carbonate for this is endogenously produced.6
As an alkalising mineral calcium is expected in supplemental alkalising formulas. However, with calcium supplements come inherent risks of accelerating vascular calcium deposition.7 Vitamin K2, in adequate levels in the circulation, can inhibit and reverse calcium deposition and vascular calcification.8 Additionally, vitamin K2 activates osteocalcin, stimulating bone mineralisation and uptake of calcium into the bone matrix.7
As in so many instances, the correct supplementation is necessary to achieve desired outcomes. Always consult a qualified Naturopath and resist the temptation to self-prescribe.
References
- Carnauba RA, Baptistella AB, Paschoal V, Hübscher GH. Diet-induced low-grade metabolic acidosis and clinical outcomes: A review. Nutrients. 2017;9(6). doi:10.3390/nu9060538
- Gregory NS, Kumar R, Stein EM, et al. Potassium citrate decreases bone resorption in postmenopausal women with osteopenia: A randomized, double-blind clinical trial. Endocr Pract. 2015;21(12):1380-1386. doi:10.4158/EP15738.OR
- Lindberg JS, Zobitz MM, Poindexter JR, Pak CYC. Magnesium bioavailability from magnesium citrate and magnesium oxide. J Am Coll Nutr. 1990;9(1):48-55. doi:10.1080/07315724.1990.10720349
- Carr AJ, Slater GJ, Gore CJ, Dawson B, Burke LM. Effect of sodium bicarbonate on [HCO3-], pH, and gastrointestinal symptoms. Int J Sport Nutr Exerc Metab. 2011;21(3):189-194. doi:10.1123/ijsnem.21.3.189
- Antacids. International Foundation for Gastrointestinal Disorders. https://www.iffgd.org/diet-treatments/antacids.html. Published 2019. Accessed May 12, 2020.
- Casiday, R & Frey R. Blood, Sweat and Buffers:pH Regulation During Exercise. General Chemistry Lab Tutorials. http://www.chemistry.wustl.edu/~edudev/LabTutorials/CourseTutorials/bb/Buffer/Buffer.htm. Published 2007. Accessed May 19, 2020.
- Kaneki M. Proper Calcium Use: Vitamin K2 as a Promoter of Bone and Cardiovascular Health. Clin Calcium. 2005;15(4):605-610.
- Nutrient reference values for Australia and New Zealand Calcium. Australian Government National Health and Medical Reseach Council. https://www.nrv.gov.au/nutrients/calcium. Published 2014.
- Abate EG, Clarke BL. Review of hypoparathyroidism. Front Endocrinol (Lausanne). 2017;7(JAN):1-7. doi:10.3389/fendo.2016.00172
